At InnoCyto, we believe powerful science begins with confidence at the bench. Our new Protocols Library brings together clear, optimized, and reproducible workflows designed to help you get the most out of our antibodies, fluorescence-labeled proteins, and cell analysis tools. Whether you’re advancing immunology, oncology, stem cell biology, or assay development, each protocol is crafted by our scientific team to deliver reliable performance and consistent results, right from the first experiment.
Explore step-by-step guides, expert tips, and application-focused best practices that streamline your experiments and elevate data quality. With InnoCyto protocols, you're not just following a method, you’re speeding your path to discovery.
Cell surface staining specifically targets proteins expressed on the cell membrane using fluorescently labeled antibodies, enabling identification and quantification of distinct cell populations.
Intracellular cytokine staining (ICS) is a flow cytometry technique using fixation and permeabilization to detect and quantify cytokines produced within cells after cell stimulation.
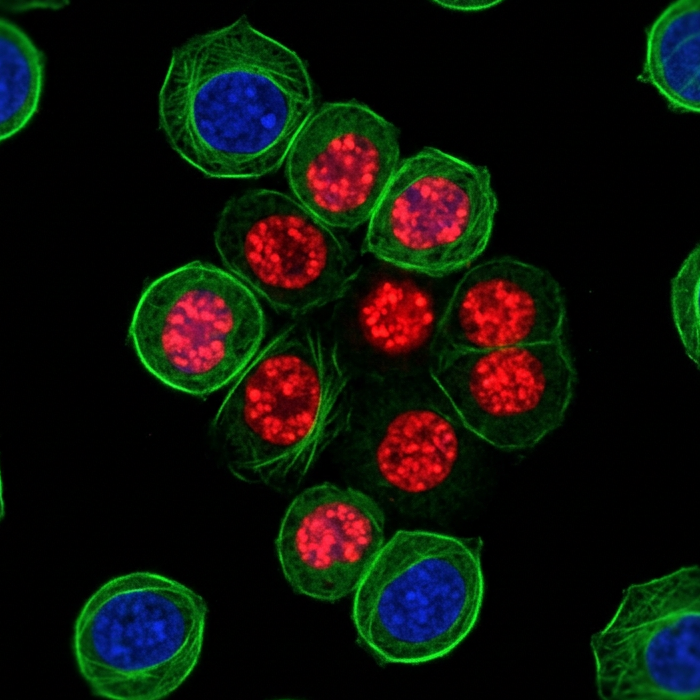
Ki-67 is a nuclear protein expressed during all active phases of the cell cycle (G1, S, G2, and M) but is absent in resting cells (G0). This protocol uses 70% ethanol fixation, which simultaneously fixes and permeabilizes cells in a single step, enabling subsequent staining of Ki-67 using antibodies. Both plate and tube-based methods are included.
The direct ELISA, one of the simplest forms of ELISA, involves immobilizing the antigen directly onto the plate and then detecting it with an enzyme-conjugated antibody specific to the antigen.
This highly sensitive sandwich ELISA protocol leverages a capture antibody and a biotinylated antibody, plus a streptavidin-enzyme conjugate to detect and quantify target proteins in complex biological samples with exceptional specificity and signal amplification. Perfect for researchers working with serum, plasma, or cell lysates, this comprehensive guide includes optimized antibody pairing strategies, troubleshooting solutions for common challenges, and expert tips for achieving publication-quality results with minimal background noise.
This streamlined sandwich ELISA protocol uses enzyme-conjugated detection antibodies for fast, reliable quantification of target proteins with reduced background and fewer steps than amplified methods. Ideal for high-throughput screening and routine analysis, this comprehensive guide delivers optimized workflows, troubleshooting solutions for conjugate-specific challenges, and expert strategies for achieving consistent results in less time than traditional indirect detection methods.
Activation of primary human T cells in vitro typically mimics physiological T-cell receptor (TCR) signaling plus co-stimulation. Engagement of CD3 provides signal 1 (TCR complex activation), while CD28 ligation supplies signal 2, promoting robust proliferation, cytokine production, and survival.
Mouse T cells can be robustly activated in vitro by engaging the T-cell receptor (TCR) complex using plate-bound anti-CD3 antibodies. Unlike human systems, many standard mouse activation protocols use anti-CD3 alone (signal 1) and rely on endogenous co-stimulatory signals from APCs or added IL-2; however, you can add soluble anti-CD28 for stronger co-stimulation if desired.

This protocol describes a straightforward method for purifying IgG antibodies or Fc- tagged proteins using Protein A agarose resin through either column-based or batch binding approaches. Antibodies are captured via high-affinity Fc–Protein A interactions, washed to remove contaminants, and eluted under low-pH conditions followed by immediate neutralization to preserve antibody integrity.

This protocol describes a step-by-step method for purifying His-tagged proteins using Nickel affinity resin through either column-based or batch binding approaches. Proteins are captured via high-affinity His-Nickel interactions, washed to remove contaminants, and eluted under low-pH conditions followed by immediate neutralization to preserve protein integrity.
This protocol describes the purification of DYKDDDDK (FLAG)-tagged proteins using 1002AH1 antibody-based affinity resin, enabling highly specific capture under native conditions. Bound proteins are recovered using DYKDDDDK peptide or low pH elution, providing flexibility for maintaining protein structure and function.

This optimized Western blot workflow integrates pre-transfer gel validation using BlinkBlue staining to improve experimental confidence and reproducibility. Incorporating rapid QC steps alongside standardized antibody and transfer conditions ensures consistent, high-quality protein detection.
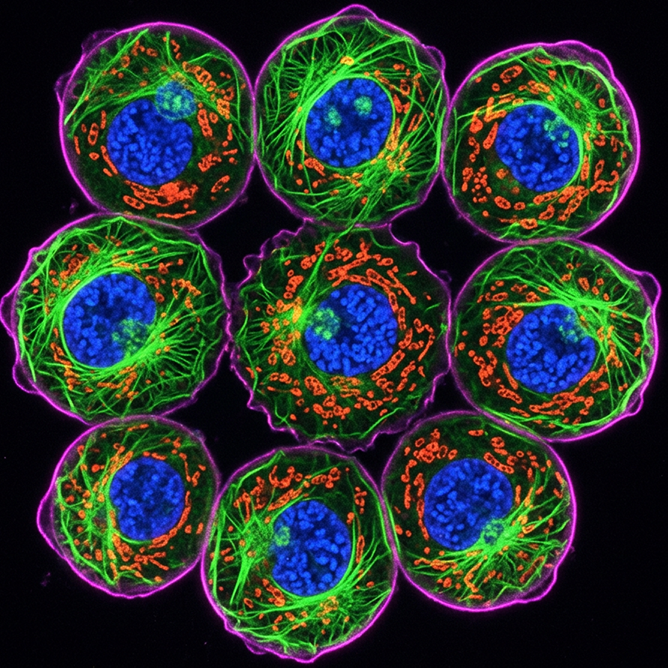
This protocol describes immunofluorescence microscopy of cell suspensions, a technique for visualizing and localizing specific proteins and cellular structures in non-adherent cells using fluorophore-conjugated antibodies. The method involves attaching cells to slides via cytospin or settling, fixing to preserve cellular architecture, permeabilizing for intracellular targets, blocking non-specific binding, and sequential antibody staining followed by fluorescence microscopy imaging. This approach is particularly valuable for examining cellular phenotypes and protein localization in suspension cells such as lymphocytes, hematopoietic cells, and cultured cell lines, providing spatial and morphological context that complements flow cytometry data.
Contact an expert for more detailed support.